IL-6 AND PMR
Understanding PMR Pathophysiology: An Expert's Perspective
Hear Dr Leonard Calabrese Discuss the Pathophysiology of PMR and the Role of Elevated IL-6 in PMR:
Listen to an expert's perspective on the following questions:
- What are the patterns of inflammation commonly seen in patients with PMR?
- What immune pathways have been linked to PMR disease activity?
- What makes the pathophysiology of PMR different from other rheumatic diseases?
- What evidence exists for the role of IL-6 in PMR pathophysiology?
- How does IL-6 contribute to disease progression?
Proinflammatory cytokines play important roles in the pathogenesis of PMR1
Key hallmarks associated with PMR, such as painful inflammation, can be attributed, in part, to certain immune cells and cytokines, such as IL-6, IL-1, IL-17, IL-10, and TNFα.1-5
![]()
![]()
IL-6 is implicated in the pathogenesis of PMR
Under normal physiologic conditions, IL-6 performs many functions, including vital proinflammatory functions in response to infection or injury.6 However, persistently elevated IL-6 levels drive inflammation, which can help promote the pathologic conditions observed in autoimmune and inflammatory conditions such as PMR.1,7,8
IL-6 impacts PMR symptoms
- IL-6 levels correlate with PMR disease activity3
- Elevated serum soluble IL-6 concentrations were identified as a potential prognostic marker for PMR relapses3,9
- Synovitis is observed in PMR as a type of inflammation accompanied by increased level of IL-6 in the synovial fluid7,10,11
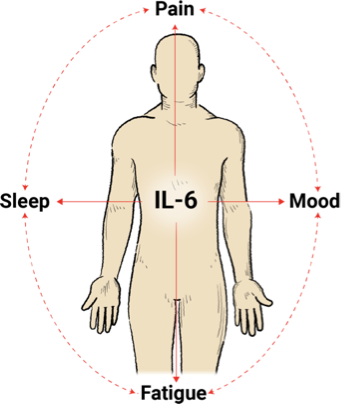
A cycle of inflammation
A study of patients with PMR demonstrated that muscle pain and stiffness concurrently developed when IL-6 concentrations were increased.1
In PMR, IL-6 is a major driver of acute-phase response and systemic inflammation and1,8:
- Stimulates increases in C-reactive protein and erythrocyte sedimentation rate4
- Plays an important role in pain perception and peripherally induced pain, and the degree of IL-6 elevation correlates with the magnitude of pain12
The increase of IL-6 is associated with fatigue, impaired sleep, and mood disorders, particularly anxiety and depression.13,14
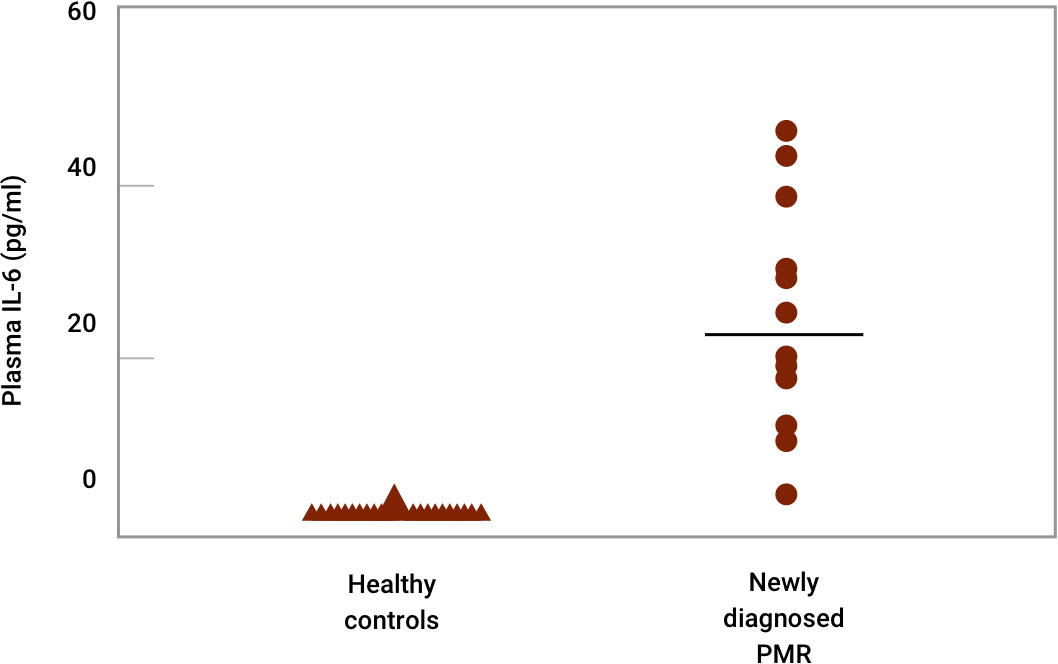
Elevated concentrations of plasma IL-6 are a characteristic feature in patients with PMR vs healthy controls3
Correlation of interleukin-6 production and disease activity in polymyalgia rheumatica and giant cell arteritis. Roche NE, Fulbright JW, Wagner AD, et al. Copyright © 1993, American College of Rheumatology. Reproduced with permission of John Wiley & Sons Ltd.
Study Design3
This study explored the role of proinflammatory cytokines in giant cell arteritis (GCA) and polymyalgia rheumatica (PMR), two clinically related syndromes characterized by an intense acute-phase reaction. In particular, to determine plasma concentrations of interleukin-6 (IL-6) and tumor necrosis factor α (TNFα) and to correlate changes in plasma IL-6 levels with clinical symptoms during corticosteroid therapy. Thirteen patients with untreated PMR and 19 patients with untreated, biopsy-proven GCA were entered in the study with twenty normal subjects who had no evidence of an inflammatory disease as controls. IL-6 and TNFα concentrations were determined in plasma samples from patients with untreated PMR or GCA, and plasma IL-6 levels were monitored in patients receiving long-term therapy (14 months) with corticosteroids. To identify IL-6–producing cells, the polymerase chain reaction was used to detect IL-6 messenger RNA. In vitro production of IL-6 and IL-2 by peripheral blood mononuclear cells (PBMC) from treated and untreated patients was quantified using IL-6– and IL-2–specific bioassay systems.
Roche NE, Fulbright JW, Wagner AD, et al. Correlation of interleukin-6 production and disease activity in polymyalgia rheumatica and giant cell arteritis. Arthritis & Rheumatism. 1993;36(9):1286-1294. Reproduced by permission of Oxford University Press on behalf of the British Society for Rheumatology.
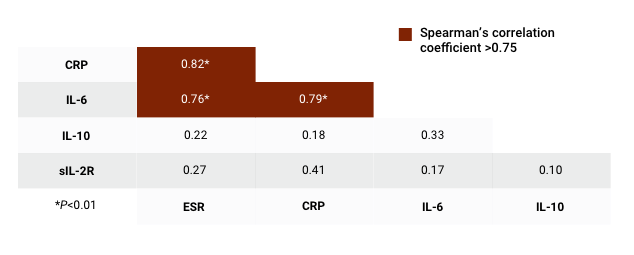
Van der Geest K, Abdulahad WH, Rutgers A, et al. Serum markers associated with disease activity in giant cell arteritis and polymyalgia rheumatica. Rheumatology. 2015;54:1397-1402, by permission of Oxford University Press on behalf of the British Society for Rheumatology.
Study Design15
This prospective study compared multiple serum markers for their ability to detect active disease in patients with GCA and/or PMR. It analyzed twenty-six markers related to immune cells that may be involved in GCA and PMR were determined by ELISA and multiplex assay in the serum of 24 newly diagnosed, untreated GCA/PMR patients, 14 corticosteroid (CS)-treated GCA/PMR patients in remission and 13 healthy controls. Receiver operating characteristic analysis with area under the curve and Spearman’s correlation coefficients were performed.
Van der Geest K, Abdulahad WH, Rutgers A, et al. Serum markers associated with disease activity in giant cell arteritis and polymyalgia rheumatica. Rheumatology. 2015;54:1397-1402, https://doi.org/10.1093/rheumatology/keu526. Reproduced by permission of Oxford University Press on behalf of the British Society for Rheumatology.
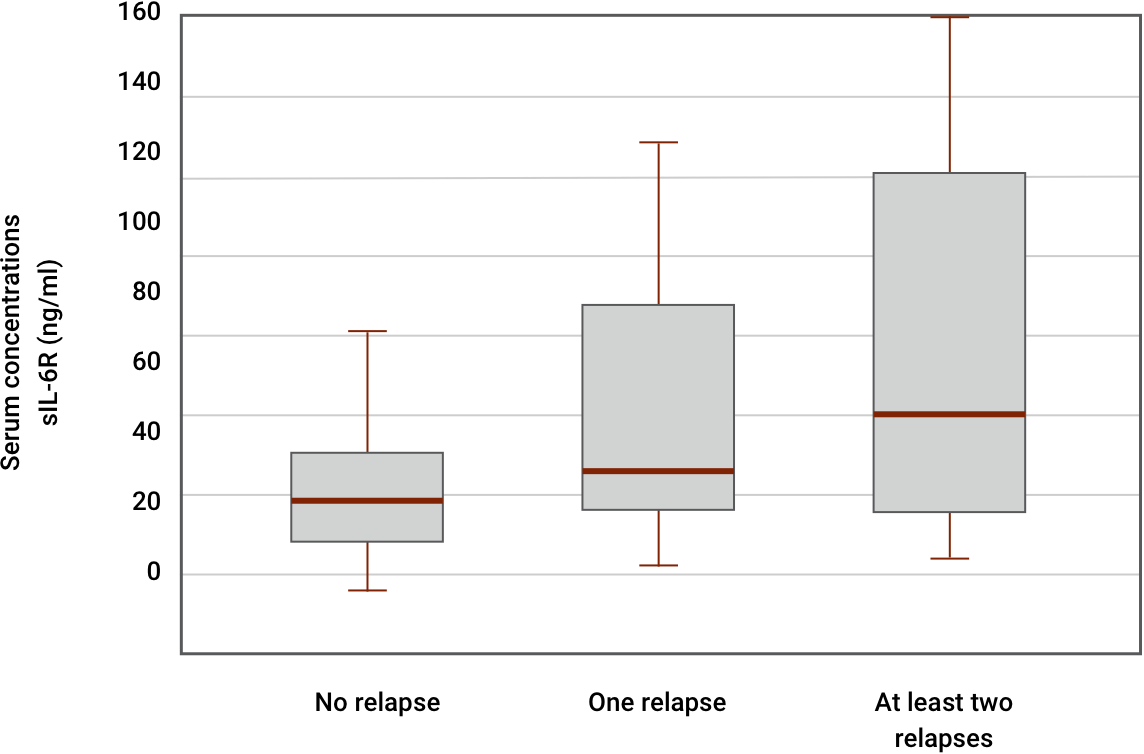
Serum interleukin-6 receptor in polymyalgia rheumatica: a potential marker of relapse/recurrence risk. Pulsatelli L, Boiardi L, Pignotti E, et al. Copyright © 2008, American College of Rheumatology. Reproduced with permission of John Wiley & Sons Ltd.
Study Design15
This study investigated the modulation of systemic levels of soluble interleukin-6 receptor (sIL-6R) and soluble gp130 (sgp130) in untreated and treated polymyalgia rheumatica (PMR) patients during a followup period of at least 24 months in order to evaluate the relationship of these molecules with clinical outcome and their feasibility to provide a prognostic tool in clinical practice. It analyzed sIL-6R and sgp130 serum levels in 93 PMR patients, and 46 age-matched normal controls, at disease onset and at 1, 3, 6, 12, and 24 months of followup during corticosteroid therapy by enzyme-linked immunosorbent assay.
Pulsatelli L, Boiardi L, Pignotti E, et al. Serum interleukin-6 receptor in polymyalgia rheumatica: a potential marker of relapse/recurrence risk. Arthritis Rheum. 2008;59(8):1147-1154, DOI 10.1002/art.23924, https://doi.org/10.1093/rheumatology/keu526. Reproduced by permission of Oxford University Press on behalf of the British Society for Rheumatology.

References: 1. Gonzalez-Gay MA, Matteson EL, Castañeda S. Polymyalgia rheumatica. Lancet. 2017; 390:1700-1712. 2. Toussirot E, Régent A, Devauchelle-Pensec V, et al. Interleukin-6: a promising target for the treatment of polymyalgia rheumatica or giant cell arteritis? RMD Open. 2016;2:e000305. doi:10.1136/rmdopen-2016-000305. 3. Roche NE, Fulbright JW, Wagner AD, et al. Correlation of interleukin-6 production and disease activity in polymyalgia rheumatica and giant cell arteritis. Arthritis & Rheumatism. 1993;36(9):1286-1294. 4. Guggino G, Ferrante A, Macaluso F, et al. Pathogenesis of polymyalgia rheumatica. Reumatismo. 2018;70(1):10-17. 5. Martinez-Taboada VM, Alvarez L, RuizSoto M, et al. Giant cell arteritis and polymyalgia rheumatica: role of cytokines in the pathogenesis and implications for treatment. Cytokine. 2008;44(2):207-220. 6. Tanaka T, Kishimoto T. Targeting interleukin-6: all the way to treat autoimmune and inflammatory diseases. Int J Biol. 2012;8(9):1227-1236. 7. Jiemy WF, Zhang A, Boots AMH, et al. Expression of interleukin-6 in synovial tissue of patients with polymyalgia rheumatica. Ann Rheum Dis. 2022;0:1-2. doi:10.1136/annrheumdis-2022-222873. 8. Salvarani C, Cantini F, Niccoli L, et al. Acute-phase reactants and the risk of relapse/recurrence in polymyalgia rheumatica: a prospective followup study. Arthritis & Rheumatism. 2005;53(1):33-38. 9. Mori S, Koga Y. Glucocorticoid-resistant polymyalgia rheumatica: pretreatment characteristics and tocilizumab therapy. Clin Rheumatol. 2016;35:1367-1375. 10. Zen-nyoji A, Shimizu H, Ohtani K, et al. Increased RAHA titer and interleukin-6 levels in the synovial fluid in a patient with polymyalgia rheumatica. Intern Med. 1993;32(6):484-486. 11. Lundberg I, Sharma A, Turesson C, Mohammed AJ. An update on polymyalgia rheumatica. J Intern Med. 2022;292:717-732. 12. Sebba A. Pain: a review of interleukin-6 and its roles in the pain of rheumatoid arthritis. Open Access Rheumatol. 2021;13:31-43. 13. Grygiel-Górniak B, Puszczewicz M. Fatigue and interleukin-6—a multi-faceted relationship. Reumatologia. 2015;53(4):207-212 14. Acharya S, Musa R. Polymyalgia Rheumatica. NCBI Bookshelf. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022. 15. Van der Geest K, Abdulahad WH, Rutgers A, et al. Serum markers associated with disease activity in giant cell arteritis and polymyalgia rheumatica. Rheumatology. 2015;54:1397-1402. 16. Pulsatelli L, Boiardi L, Pignotti E, et al. Serum interleukin-6 receptor in polymyalgia rheumatica: a potential marker of relapse/recurrence risk. Arthritis Rheum. 2008;59(8):1147-1154.